Chemical Dominoes
For this project our objective was to combine our knowledge of chemical reactions with simple physics concepts to create a Rube Goldberg Machine. My group and I decided to turn on an LED light as our final step to our chemical dominoes. These chemical reactions included combustion reactions, synthesis reactions, double displacement reactions, and acid base reactions.
Combustion Reaction: occurs when a reactant combines with oxygen to produce oxides of all other elements as products
Synthesis Reaction: two or more substances combine to form a new compound (A+B=C)
Double-Displacement Reaction: two compounds as reactants and two different compounds as products
Acid-Base Reaction: When an acid and a base are placed together, they react to neutralize the acid and base propertied, producing a salt.
Combustion Reaction: occurs when a reactant combines with oxygen to produce oxides of all other elements as products
Synthesis Reaction: two or more substances combine to form a new compound (A+B=C)
Double-Displacement Reaction: two compounds as reactants and two different compounds as products
Acid-Base Reaction: When an acid and a base are placed together, they react to neutralize the acid and base propertied, producing a salt.
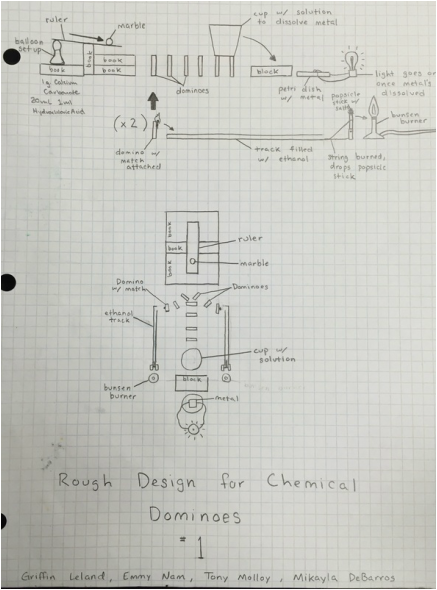
Blueprint:
Below is an original blueprint of our chemical dominoes
Below is an original blueprint of our chemical dominoes
Chemistry Concepts:
1). Carbon Dioxide Balloon
Materials:
- Hydrochloric acid
- Calcium Chloride
2g of CaCl2 is poured into a balloon. We then capped the CaCl2-filled balloon to cover a flask containing 10ml of hydrochloric acid. The reaction of the two combines to create CO2, which then causes the balloon to rise. We place the balloon under a ruler to act as a lever, making a marble roll down to continue the operation.
Formula: 2 HCl +Ca(OH)3 ---> CaCl2 + CO2 + H2O
2). Alcohol Burning
Materials:
- Alcohol (liquid)
- Heat source (a lit match)
Reaction of alcohol and fire, burns through a string, releasing a popsicle-stick with copper chloride into a bunsen burner. Alcohol provides as a instigator to the flame and allowing the heat to burn our string.
Formula: C2H6O + 3(O2) ---> 3(H2O) + 3(CO2)
3). Produce Colored Flames
Materials:
- Popsicle-stick
- Copper Chloride (powder)
- Bunsen Burner
- Water
- String
We dipped the tip of the popscile stick into water to allow the copper chloride to stick to it better. Then we set it into place in between two blocks held by a string, and suspended over a bunsen burner. Once the copper chloride come in contact with the flame, a streak of light teal appears as a reaction. This occurs because the salts of the CuCl2 are heated, raising the electrons to a new energy level, and creating a new colored flame.
Formula: CuCl2 + Energy --> Emittance of Photons
4). Disintegrating Metal
Materials:
- Aluminum foil
- Circuit board (battery, resistors, LED, etc.)
- Watch glass
- Copper Chloride (liquid)
Through a funnel, we poured CuCl2 to disintegrate the aluminum foil that we placed in a watch glass. Once the CuCl2 destroyed the metal, the light turned on. There were also very small traces of copper left behind. This was an example of a single replacement reaction and the finishing step of our project.
Here is our chemical dominoes in action
1). Carbon Dioxide Balloon
Materials:
- Hydrochloric acid
- Calcium Chloride
2g of CaCl2 is poured into a balloon. We then capped the CaCl2-filled balloon to cover a flask containing 10ml of hydrochloric acid. The reaction of the two combines to create CO2, which then causes the balloon to rise. We place the balloon under a ruler to act as a lever, making a marble roll down to continue the operation.
Formula: 2 HCl +Ca(OH)3 ---> CaCl2 + CO2 + H2O
2). Alcohol Burning
Materials:
- Alcohol (liquid)
- Heat source (a lit match)
Reaction of alcohol and fire, burns through a string, releasing a popsicle-stick with copper chloride into a bunsen burner. Alcohol provides as a instigator to the flame and allowing the heat to burn our string.
Formula: C2H6O + 3(O2) ---> 3(H2O) + 3(CO2)
3). Produce Colored Flames
Materials:
- Popsicle-stick
- Copper Chloride (powder)
- Bunsen Burner
- Water
- String
We dipped the tip of the popscile stick into water to allow the copper chloride to stick to it better. Then we set it into place in between two blocks held by a string, and suspended over a bunsen burner. Once the copper chloride come in contact with the flame, a streak of light teal appears as a reaction. This occurs because the salts of the CuCl2 are heated, raising the electrons to a new energy level, and creating a new colored flame.
Formula: CuCl2 + Energy --> Emittance of Photons
4). Disintegrating Metal
Materials:
- Aluminum foil
- Circuit board (battery, resistors, LED, etc.)
- Watch glass
- Copper Chloride (liquid)
Through a funnel, we poured CuCl2 to disintegrate the aluminum foil that we placed in a watch glass. Once the CuCl2 destroyed the metal, the light turned on. There were also very small traces of copper left behind. This was an example of a single replacement reaction and the finishing step of our project.
Here is our chemical dominoes in action
Reflection:
For this project, our group definitely had several setbacks, but we managed to overcome them and deliver a successful product as a result. A few mishaps included the inconsistency of our dominoes, our cup full of solution missing the landing mark, and the matches not staying lit. However, we did our best to fix these blunders and stay united as a group. Although we did not get our chemical dominoes to fully work on film, we are still confident that with a few tweaks it should function properly.
Throughout this process, I came to learn a few things about myself and gained a new understanding about chemical reactions and their properties. For one, I discovered that I grasp concepts better through observing the correct way to do it first, then trying it on my own. For example, seeing Emmy set up the calcium chloride balloon the first time, allowed me to understand how to do it for myself. Secondly, I realized that I provide my team with some creative ideas and by sharing more of my voice should better how groups overall communication and performance.
A couple things I look to improve on in future projects are being more active and hands on, as well as keeping my group and myself on task. No matter if I take on the leadership role in a certain project or not, it's still important to remain hands on with the project and always contributing. This is something I definitely want to work on in the future. Staying on task is key in any group work and by managing time should allow for a better product in the end.
Recap:
2 Pits - time management and inconsistency of some of our operations
2 Peaks - determination in getting our chemical dominoes to work and good communication with no arguments
For this project, our group definitely had several setbacks, but we managed to overcome them and deliver a successful product as a result. A few mishaps included the inconsistency of our dominoes, our cup full of solution missing the landing mark, and the matches not staying lit. However, we did our best to fix these blunders and stay united as a group. Although we did not get our chemical dominoes to fully work on film, we are still confident that with a few tweaks it should function properly.
Throughout this process, I came to learn a few things about myself and gained a new understanding about chemical reactions and their properties. For one, I discovered that I grasp concepts better through observing the correct way to do it first, then trying it on my own. For example, seeing Emmy set up the calcium chloride balloon the first time, allowed me to understand how to do it for myself. Secondly, I realized that I provide my team with some creative ideas and by sharing more of my voice should better how groups overall communication and performance.
A couple things I look to improve on in future projects are being more active and hands on, as well as keeping my group and myself on task. No matter if I take on the leadership role in a certain project or not, it's still important to remain hands on with the project and always contributing. This is something I definitely want to work on in the future. Staying on task is key in any group work and by managing time should allow for a better product in the end.
Recap:
2 Pits - time management and inconsistency of some of our operations
2 Peaks - determination in getting our chemical dominoes to work and good communication with no arguments